Biography
In 1998 I was awarded my master’s degree with honours in Pharmaceutical Chemistry and Technology by the University of Padua, where I specialized in biotechnology (this is equivalent to a combined BPh and MPh in the UK system). After a brief period of undergraduate research, externally funded, I moved to work as a researcher at the Siena vaccine-hub – Research Center of the Chiron Vaccines (now GSK’s research centre). I was involved in vaccine and antimicrobials discovery and development, including Streptococcus pneumoniae and Neisseria meningitidis. After this company experience, I obtained my PhD in Cellular and Molecular Biology and Pathology at the University of Padua, working on host-pathogen interactions and microbial virulence factors regarding Helicobacter pylori. For my post-doctoral formation I joined Prof D Goldberg’s lab in Washington University in St Loui, MO, working on malaria, field where my lab then specialized. Awarded an ERC-Reintegration Grant I moved back to Europe, where, after a brief experience at the University of Perugia, I moved to the University of Manchester in 2013 supported by a transition-to independence bursary. I was then offered an Assistant professorship position back in USA in 2017, where I was enabled to perform cutting-edge research regarding cerebral malaria onset using the in vitro BBB and the animal model in collaboration with Prof Goldberg. While considering the possibility to establish my research in USA, in 2018, I was appointed Lecturer in the School of Medicine at Keele University where, since 2019, I am trying (despite the difficulties of COVID19-pandemic) to start up my independent lab in the UK, working on Plasmodium cell biology and host-pathogen interaction.
I have actively worked in medical research, focusing on infectious diseases, encompassing both lab basic research and outreach field activities. I have expertise in vaccines, drugs and targets intersecting the fields of malaria, haematology, bacteriology, virulence factors, and immunology. Expertise in these areas where matured working in top class laboratories in both academics and companies under the direction of exceptional scientists: A Fontana (organic chemist); E De Filippis (protein chemist); E Di Cera (master of enzymology); R Rappuoli (pioneer of reverse vaccinology); C Montecucco (PhD supervisor); DE Goldberg (a world-wide renowned malariologist); C Thompson, I Roberts and M White (in Manchester).
My major contributions are the characterization of two novel antimalarial targets Calpain and PlasmepsinV that disclosed new pieces of parasite biology, followed by numerous other discoveries and the establishment of a set of biotechnology tools to move forward genetic, enzymology and immune research in malaria and immunology. My published work is in top rated journals such as Science, PNAS, Molecular Microbiology, Nature.
My current research regards the study of Plasmodium species, causative agents of human malaria studying their cell biology aiming to:
- validating targets for novel therapeutic options;
- enabling innovative vaccination strategies: and
- dissecting the virulence factors affecting the immune responses and haematology barriers, that are mostly affected by malaria infection, such as the Blood Brain Barrier (BBB).
Strong expertise in the lab, always devoted in pushing the boundaries of biotechnology solutions to the study of Plasmodium, underpins all the projects and preferentially uses in-vitro models. Recently, I joined efforts with the Goldberg lab to study Plasmodium virulence factors involved in the development of cerebral malaria. Via collaboration with DRC-doctors and scientists, I am also leading small projects with impact on health and social benefit in the remit of infection diseases’ interventions in the area of Katanga (DRC).
I have been an ordinary member of the council for the British Society of Parasitology starting from 2019, a member of the editorial board for Enliven: Immunology and Immunotechniques and Advances in Infectious Diseases & Therapy. I am also a member of CAEP, Midland Immunology group, as well as, the Academic Women Network @Washington University in St. Louis; the Expert Women in Life Sciences – WILS database; the Italian Malaria Network – CIRM; the Marie Curie Fellows Association – MCFA; (the former) Outreach Evimalar task-force; the American Society of Biochemistry and Molecular Biology (ASBMB); and the WiM (Woman in Malaria network).
For more information, please, feel free to email me for a chat or visit my webpages at:
- ORCIDhttps://orcid.org/0000-0002-2269-7078
- ResearcherID D-4674-2014
- Scopus Author ID 35861853300
- Research gate Ilaria Russo
- Linked-in Ilaria Russo
- FdS org Ilaria Russo (for extra-academic outreach activities)
Research and scholarship
Research background
Apicomplexan Plasmodium parasites are the causative agents of malaria, one of the major killers among infectious diseases. Containment measures have been implemented. However, malaria morbidity and mortality have reached a plateau in the past five years and, in some instances, even starting back to increase in some countries. Multiple factors hinder the disease control's effectiveness, among which rising drug and insecticide resistance, weak surveillance, a poorly performing vaccine and decreased availability of funding (WHO-Malaria Report-2020). New approaches to control malaria are therefore urgently needed.
My laboratory focuses on research projects that respond to the call for novel validated molecular malaria targets and vaccinations, both priorities identified under the Global Action Malaria plan and the Millennium Goals.
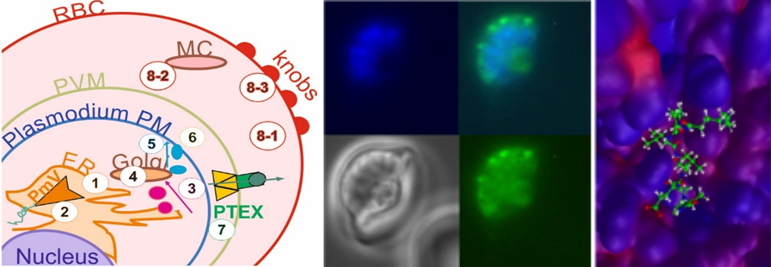
Above: a model of Plasmodium secretion; live fluorescent DPAP3-sporozoites; 3D-docking of PMV-inhibitor
Past and current endeavours
In over-15-years, my work on Plasmodium falciparum, the deadliest strain causing human malaria, has provided and continues to provide great contributions in cell biology, genetics, biotechnology approaches and biochemistry. Well-known for cutting-edge discoveries into malaria cell biology and pathology, my research aims to tackle urgent unmet medical needs affecting malaria with a strong drive to translational efforts, aiming to drug-target validation (via cell biology studies), host-pathogen interactions and vaccine development (see active projects).
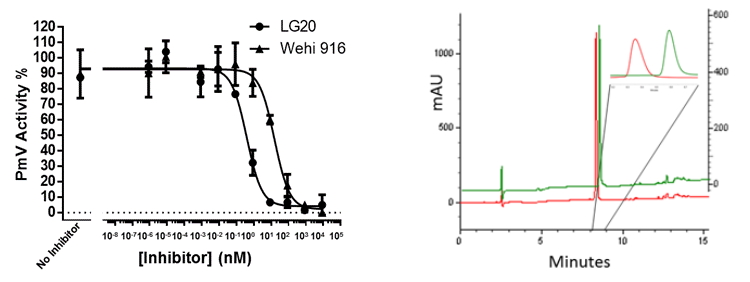
My technical skills and expertise have allowed me to tackle highly challenging tasks and projects that delivered hard-to-achieve results and breakthroughs, such as the discovery and characterization of Pf_Calpain and Pf_PlasmepsinV (published in Nature, PNAS, Molecular Microbiology and Science – see publications). I have also published important technological advancements, designing and validating new techniques, such as the first small-molecule-driven conditional knockdown in Plasmodium, and high-throughput screening capabilities for specific cellular events generating ad hoc cell reporters (Russo,I et al, PNAS 2009; Gambini L et al. PLOS ONE 2015).
I am currently studying novel antimalarial drugs and targets, investigating the asexual cell cycle regulation and the host-pathogen interactions involved in immune responses and malaria pathogenesis.
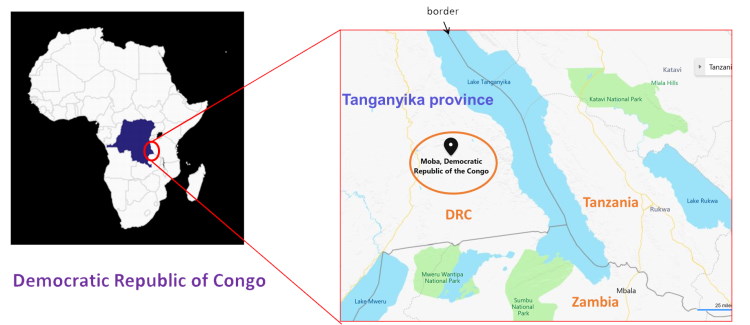
I am also operating in the Democratic Republic of Congo in collaboration with Fonte di Speranza Foundation. Working together we have established CHK, a point-of-care completed by the first diagnostic centre in Moba (Katanga region). This is a quickly growing institute offering potential strategic value for institutions, such as the WHO.
Some projects have also generated links with bio-tech companies, such as Streambio and others, that offer development and translation for lab innovations with potential in diagnostics and vaccines.
My lab has also the support of esteemed collaborators which expertise in OMICS data analyses (imaging, phenotypic readouts, chemical libraries, next-generation sequencing), high-end bioinformatics, and nanoparticles, we use in our biotechnology endeavours.
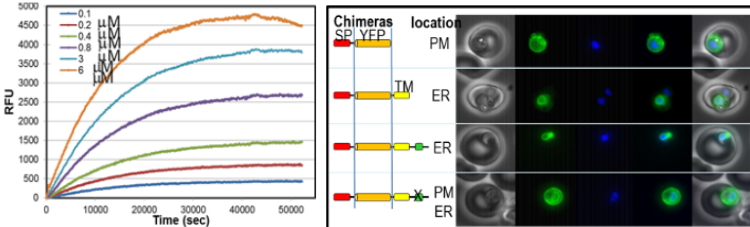
Active projects, in which students are welcomed to join our efforts, are:
- Innovative cellular assays for studying regulatory events of the asexual parasite cell cycle via chemo-genomics analysis forward and reverse genetics
- Molecular Studies on Plasmodium protein secretion, an indispensable process through which Plasmodium remodels and hijacks host cells to support its survival and growth.
- Engineering of membrane nanovesicles as innovative malaria vaccination delivering multiple antigens targeting multiple stages of the infection
- Study of the pathogenesis due to Histidine rich protein 2 and 3, Plasmodium secreted factors with exceptional structural and functional features, to validate malaria immunotherapies;
- Epidemiology and clinical studies of infectious diseases and their related policies in the Katanga region of DRC;
- Investigation of Plasmodium virulence factors affecting immune responses and hematologic barriers;
- Study of cellular homeostasis related to the maintenance of Plasmodium falciparum exceptional production of poly-Asparagine
- Design and validation of innovative molecular approaches, including genes and proteins’ modification to support novel high throughput and OMICs studies in Plasmodium falciparum.
Teaching
I am responsible for the design and delivery of a range of activities and their assessment in support of the undergraduate and post-graduate courses within the School of Medicine (Faculty of Medicine and Health Sciences) and the School of Life Sciences (PBL, Lectures, seminars, Practical classes, experiential learning and research project placements). I liaise with the teaching committee and often involved in discussions for innovations in the applied methodology and/or development of curriculum or assessments.
In 2020, I have been awarded Higher Education Academy Fellow.
Occasionally I am/was involved as internal and external viva examinations, international courses and workshops, and I regularly support the training and teaching of the staff working in the CHK diagnostic laboratory, in DRC.
The School of Medicine:
In particular, I am involved in multiple teaching formats and activities encompassing the overarching theme of Pharmacology and Microbiology, for medical students and PAs.
I also offer a number of short wet and dry projects, in high demand, for visiting, Intercalating, Inspire and ASPIRE students
The School of Life Sciences:
I offer various research projects (at the bench and in remote) for final year students: Level 6 Biology of Disease, MSc Biomedical - Blood Sciences and Medical Microbiology.
Further information
Vision and ethos

Above: From the bench - to the bedside
My job has always been conducted with contagious enthusiasm and dedication.
My vision and ethos are embedded in Keele University’s values and principles, among which I particularly feel my own and actively promote Equality, Diversity and Inclusion principles (EDI) both in the working context and in society.
I have been all my life a cooperant with NGOs promoting health, education and civil rights in Low/Middle-Countries, including DRC, Sierra Leone and Tanzania. Either for scientific or other projects, my approach in these locations is utmost respect of the cultural differences and equalitarian recognition and treatment of people I work with; and my aim and motto for all my projects in the field is Empowering Local People. If you are interested in knowing more of my external (non-research-related) activities in the field, please, visit Fonte di Speranza Foundation – web site.
I also strongly support the minimization of the animal models by continuously implementing in vitro models, such as organoids and organs-on-chips.
If private funders and charities, in line with these values, are interested in supporting my research endeavours, please, do not hesitate to get in contact and discuss possibilities at i.russo@keele.ac.uk. Valid offers will be then scrutinized for due diligence by Keele University – Research Office.
Further Information
Declared additional affiliations:
At present, I hold an honorary position as Adjunct Professor of Dept. of Internal Medicine –Infectious Diseases at Washington University in St. Louis, MO, USA and I am the CEO of DSP limited, a young company for scientific consultancies.
For home and foreign students interested in the lab activities:
I have records of high-rate satisfaction of students, the majority of which has successfully moved on in their career with success, securing both academic and company positions.
Some of them have become principal investigators, staff at GSK or in other institutions, moved to the public sector or secured prestigious fellowships in their own countries for PhD.
The training offered by the lab is generally specific to the project in which students and staff are involved. Nevertheless, the core training covers molecular biology (including CRISPR/Cas9 editing, recombineering, and NG-sequencing), cell-biology, and multi-parametrical analyses (via microscopy, flow cytometry, and phenotypic assays), biochemistry and physicochemical methodologies, basic statistical analysis, and bioinformatics.
Publications
Please note that Russo’s work is all publicly available, except for the period spent working at the company Chiron Vaccine (now GSK), due to intellectual property constraints (affecting the track record in 2003-2008); and for maternity, health and administrative issues (affecting the years 2011-2014).
Publications
- Giurisato E, Lonardi S, Telfer B, Lussoso S, Risa B, Zhang J, Russo I, et al. – The Extracellular-Regulated protein Kinase 5 is required for mediating macrophage proliferation associated with development of malignant tumors – Cancer Res ‘ June 19 2020 DOI: 10.1158/0008-5472.CAN-19-2416
- Pool MR & Russo I - The perplexing PEXEL protein secretory pathway - Nat Microbiol. 2018 Sep;3(9):969-970. doi: 10.1038/s41564-018-0235-2
- Lehmann C, DeVries L, Tan S, Russo I, Sanchez-Lopez M, Goldberg DE, Deu E - Plasmodium falciparum dipeptidyl aminopeptidase 3 (DPAP3) activity is important for efficient erythrocyte invasion –– Plos Pathogen 2018 May 16;14(5):e1007031. doi: 10.1371/journal.ppat.1007031
- Alnabulsi S, Hussein B, Santina E, Alsalahat I, Kadirvel M, Magwaza RN, Bryce RA, Schwalbe CH, Baldwin AG, Russo I, Stratford IJ & Freeman S - Evaluation of analogues of furan-amidines as inhibitors of NQO2 for potential use in the treatment of cancer and malaria - Bioorganic & Medicinal Chemistry Letters – 20182018 - 28, 8, 1292-1297 – doi:10.1016/j.bmcl.2018.03.025
- Giurisato E, Telfer B, Lonardi S, Wang J, Russo I, Gray NS, Vermi W, Tournier C - Myeloid ERK5 deficiency suppresses tumor growth by blocking pro-tumor macrophage polarization via STAT3 inhibition – PNAS 2018
- Nasamu SA, Glushakova S, Russo I, Vaupel B, Oksman A, Kim AS, Fremont DH, Tolia N, Beck JR, Meyers MJ, Niles JC, Zimmerberg J, Goldberg DE - Plasmepsins IX and X are essential and druggable mediators of malaria parasite egress and invasion – Science 2017
- Alnabulsi S, Santinaa E, Russo I, Kadirvel M, Chadwick A, Bichenkova E, Bryce R, Nolan K, Demonacos C, Stratford I and Freeman S - Furan-amidines as potent NRH: Quinone Oxidoreductase 2 (NQO2) inhibitors: Design, Synthesis and Biological Evaluation –European Journal of Medicinal Chemistry 111 (2016) 33e45.
- Gambini L×,Rizzi L, Pedretti A, Taglialatela-Scafati O, Carucci M, Pancotti A, Galli C, Read M, Giurisato E, Romeo S & Russo I×, - Picomolar inhibition of Plasmepsin V, an essential Malaria protease, achieved exploiting the prime region - ×Equally contributed – Plos One 11/2015; 10(11):e0142509. DOI:10.1371/journal.pone.0142509.
- Moretti E, Collodel G, Mazzi L, Russo I & Giurisato E - Ultrastructural study of spermatogenesis in KRS2 deficient mice - Transgenic Research – 2015 Aug;24(4):741-51.
- P. falciparum & P. vivax Novel Targets & Classes – final report of CRIMALDDI workshop No1 – October 2010 - Russo I among the participants/authors – published on line at web link
- Russo I, Babbitt S, Muralidharan V, Butler T, Oksman A and Goldberg DE – Plasmepsin V licenses Plasmodium proteins for export into the host erythrocyte – Article in Nature 2010 Feb 4; 463(7281):632-636.(web)
- Fennell C, Babbitt S, Russo I, Wilkes J, Ranford-Cartwright L, Goldberg DE, Doerig C–PfeIK1, a eukaryotic initiation factor 2 kinase of the human malaria parasite P. falciparum, regulates stress-response to amino-acid starvation – Malaria Journal 2009 May 12;8:99.web
- Russo I., Oksman A. and Goldberg DE – Fatty Acid Acylation Regulates Trafficking of the Unusual P. Falciparum Calpain to the Nucleolus– Molecular Microbiology; 2009 Apr; 72(1):229-45. Epub: Feb 23 2009. web
- Russo I., Oksman A., Vaupel B and Goldberg DE – A calpain unique to alveolates is essential in P. falciparum and its knockdown reveals an involvement in pre-S-phase development– Proc Nat Acad of Science; 2009 Feb 3;106(5):1554-9. Epub 2009 Jan 22. web -
- De Filippis, V., Colombo, G., Russo, I., Spadari, B. and Fontana, A. –Probing the Hirudin-Thrombin Interaction by Incorporation of Non-coded Amino Acids and Molecular Dynamics Simulation - Biochemistry; 2002; 41(46); 13556-13569. web
- De Filippis, V; Russo, I; Salmaso, S; Fontana, A - Protein engineering by non-coded amino acids - : Italian Journal of Biochemistry - Volume: 49 Issue: 1 - MAR 2000
- De Filippis, V., Russo, I., Vindigni, A., Di Cera, E. and Fontana, A. -(1999)- A highly potent and selective thrombin inhibitor obtained by cumulative incorporation of non-natural amino acids into the N-terminal domain 1-47 of hirudin - Protein Science, 1999, 8, 2213-2217. web
Books
(not peer-reviewed)
- Russo I, & Goldberg DE - Plasmepsin V. In Handbook of Proteolytic Enzymes, 3rd ed, 2012, Woessner JF, Rawlings ND (eds), Elsevier, Oxford.
Funding
- 2021 – Royal Society Research grant – Shortlisted for funding – currently on waiting list
- 2020 – (undisclosed as under evaluation)
- 2019 – Keele University – start-up funds – new investigator award for laboratory start-up duration 2 years
- 2019 – Keele University – PhD studentship for 3 years.
- 2017 – MMV Collaborative grant - Discovery and development of drugs targeting PlasmepsinV and the PexEl secretion in Plasmodium parasites – 08/2017 – 07/2018 - Completed
- 2015-2018 Marie Curie International Reintegration Grant - Grant Agreement n° PIRG07-GA-2010-268441 -Call FP7-PEOPLE-2010-RG Proposal n°268441 – Principal investigator Ilaria Russo (single beneficiary)- Research Executive Agency of European Union (seventh framework programme) - -“Genetic High Throughput Screenings by random mutagenesis to identify Plasmodium falciparum critical genes for asexual growth, sexual differentiation and virulence affecting host immune responses” . Completed
- 04/2013 – 09/2016 Stepping Stone Fellowship. Supported by the Wellcome Trust Fund. Faculty of Life Sciences, University of Manchester, UK. Completed
- 2005-2008 - NIH Grant AI-047798. – Principal investigator: Daniel E. Goldberg - National Institutes of Health (USA)-– “Characterization of Proplasmepsin Maturase” – Aims and tasks were successfully accomplished as published. Completed.
- 2005 – Fellowship on Infectious Diseases Infectious Diseases Scholar Program(IDSP) – Beneficiary: Ilaria Russo – Dept of Medicine – Infectious disease at Washington University School of Medicine – St. Louis – MO - USA. Completed.
- 1999 – Funds for short term visit at Washington University from the Interuniversity Biotechnologies Consortium - ~Liras 4.000.000 – December 1998 – Trieste – Italy.
- 1998 – 1999 – Fellowship for Undergraduates from the Consortium of Padua Researches for purification and structural analysis of protein of biotechnological interest – Padua – Italy
A great thank you for enabling my research work to all:

Graduate-students and post-graduates
I welcome applications from students who wish to undertake PhD or MPhil postgraduate research studies in my team, as well as post-graduates. Interested students can contact me directly at i.russo@keele.ac.uk to discuss potential projects as well as the application process for the postgraduate degrees. Post-doc candidates are invited to present themselves at the same address with CV and future plans, including funding engagement.
Please, note that students and post-docs wishing to apply for funded opportunities (eg. Commonwealth Scholarship Scheme, Wellcome international funding) should contact me well in advance of the deadline in order to effectively support their application.
All backgrounds are equally welcomed, but I particularly encourage women and any other talented and motivated person to make them known, including from the BAME community, foreigners and any other minority.
Former, and current, postgraduate students include UK nationals as well as overseas students from Asia, the Middle East and Sub-Saharan Africa, undertaking both full-time and part-time studies in my laboratory.
My current research interests are indicated on the Research and Scholarship tab. Previous postgraduate students have submitted theses and published their work. Often, our research is in collaboration with international academic partners and Biotech companies
My team works closely with those of other investigators interested in parasite biology as part of the Centre for Applied Entomology and Parasitology (CAEP) providing a stimulating collegiate environment for postgraduate research students, regular contact with your supervisory team and access to modern research facilities.
Thank you for your interest in my research team.
School of Medicine
David Weatherall building
University Road
Keele University
Staffordshire
ST5 5BG
Tel: +44 (0) 1782 733937
Email: fmhs.facilities@keele.ac.uk
Admissions enquiries: enquiries@keele.ac.uk
The Clinical Education Centre
Keele University
Clinical Education Centre
University Hospitals of North Midlands NHS Trust
Newcastle Road
Stoke-on-Trent
Staffordshire
ST4 6QG
Email us: services.cecreception@keele.ac.uk
CEC general enquires: 01782 731876




