Researchers develop polymer which evolves into a helical structure
A team including a Keele researcher has developed a synthetic polymer which naturally evolves into a stronger helical structure which is commonly seen in nature, and which could help scientists create more adaptable and customisable materials.
The twisting helical shape is a structure that is commonly found in organic molecules, most famously in the double helix of a DNA molecule. It is an important shape as it helps biological molecules adapt to different environments and perform different functions.
For example, by changing how tightly they twist or even the direction of their twist, biological systems can respond and adapt to their environment, helping proteins adjust their shapes to fold correctly and perform essential tasks.
The team led by Chiba University in Japan, including Keele’s Dr Martin Hollamby and colleagues from Japan’s Shizuoka University, Kanazawa University, and Ritsumeikan University, were inspired by this structure to develop a chlorophyll-based supramolecular polymer which gradually evolves over time, from nonhelical fibres into a well-defined helical structure.
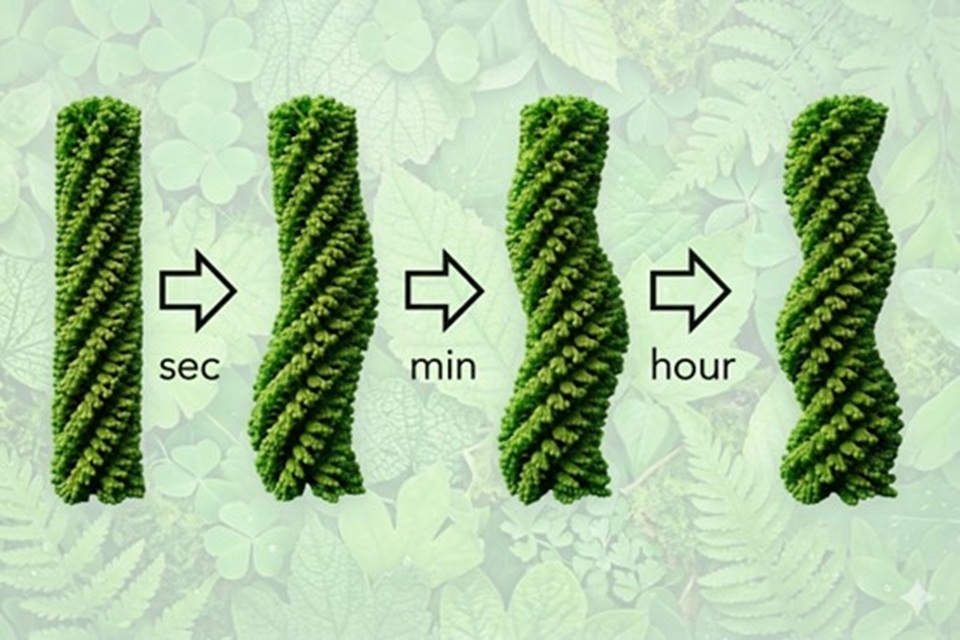
Their findings, published in the Journal of the American Chemical Society, show that the molecule they’ve developed evolves step by step, passing through several intermediate stages before reaching its final helical form. It begins as a nonhelical fibre (HF1), then develops two loose helices (HF2), and finally tightens into a more twisted structure (HF3).
Dr Hollamby at Keele led small-angle scattering experiments at ISIS, STFC, that tracked how these structures evolved over time. Starting with a solution dominated by nonhelical fibres, the team observed a gradual transformation into helical structures over the course of several days. Within the first 30 minutes, most nonhelical fibres disappeared, giving way to HF1 and HF2. Over the next few hours, HF1 was converted almost entirely into HF2. The final transformation, from HF2 to the most tightly twisted form, HF3, occurred much more slowly, taking several days.
Dr Hollamby said: “Small-angle scattering allowed us to watch changes in real-time, and crucially in their natural solution state – which is very hard to do by any other means. It’s great to see this powerful technique applied to such a unique and important supramolecular system.”
The findings point to a blueprint for designing dynamic helical structures. By creating molecular building blocks that can adopt multiple stable arrangements with only small energy differences, it may be possible to design materials that change their structure over time in a controlled way.
Most read
- Keele Innovation District moves one step closer with backing of Staffordshire local authorities
- Keele opens new insectary to advance malaria and pest control research
- Business and community leader Carol Shanahan named new Keele University Chancellor
- One‑week radiotherapy course shown to be safe and effective in the long term for early‑stage breast cancer
- Researchers find evidence of red dwarf stars “eating” their planets
Contact us
Andy Cain,
Media Relations Manager
+44 1782 733857
Abby Swift,
Senior Communications Officer
+44 1782 734925
Adam Blakeman,
Press Officer
+44 7775 033274
Ashleigh Williams,
Senior Internal Communications Officer
Strategic Communications and Brand news@keele.ac.uk.



